Article requires registration
Simple, Efficient, Sustainable What Does “Green Chromatography” Yield?
Subjects, such as resource optimization or better energy usage, have arrived in the laboratory. What does this mean for the most important separation method, chromatography? An inventory shows the amount of optimization potential in the technology that is more than 100 years old.
Related Vendors
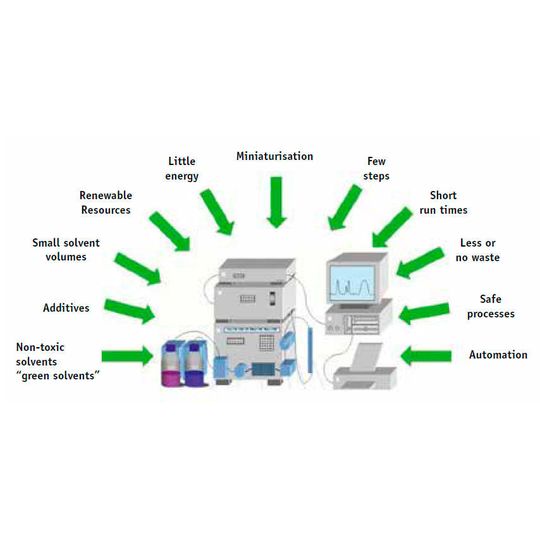
What are the goals of sustainable chromatography? First of all, energy and resource consumption are to be minimized over the entire workflow and non-toxic reagents should be used — coming from renewable ressource, whenever possible (see Fig. 1).
Green solvent
One of the most important measures is the use of non-toxic solvents. In particular, the routine operation has high consumption with an estimated usage of 1 - 1.5 litres of solvent per day. According to an estimation, which dates back to 2011 and emanates from 13 000 HPLC-devices throughout the world, this makes a consumption of 34 million liters of solvent per year [4]. Acetonitrile is the most popular solvent in standard HPLC applications due to its low viscosity. However, according to environmental and health criteria, it is critically evaluated. Though, which other possibilities are available? The use of alternative non-toxic solvents is first priority. “Solvent selection guides” classify solvents according to EHS criteria [5] and serve as an orientation aid in the selection of alternatives. Ethanol, ethylacetate or even water is frequently used and there is a large number of examples available in the literature [5]. The use of ethanol was frequently limited because of its high viscosity. In UHPLC systems, this is no longer a problem, since this withstands a higher backpressure. By raising the temperature, the viscosity of ethanol can be decreased and thus can be used in a standard HPLC-system without increasing its backpressure [6]. Another possibility to change solvent properties exists in the addition of additives. For the separation of an alkaloid mixture, a methanol:water mixture was used with a proportion of 70% methanol in the beginning. By adding cyclodextrine, the methanol content could be reduced from 70% to 50% in the first step, in the second step methanol was completely replaced by ethanol without changing the separation efficiency [7]. The so-called “Ionic Liquids” are often mentioned as “Green Solvents” in this context and are used as mobile phase additives or extraction solvents in sample preparation. Based on their low vapor pressure as well as their thermal and chemical stability, ionic liquids are classified as non-hazardous solvents and no special safety criteria have to be considered. However, the environmental effects are less studied: some decompose well, others are persistent and accumulate in the environment. On account of this, the use should be weighed despite of all factors and, if possible, compared with those with known biodegradability [8]. Other non-toxic and environmentally friendly alternatives are supercritical CO2 or supercritical water. These are used as mobile phases or in sample preparation.
Sign in or register and read on
Please log in or register and read this article. To be able to read this article in full, you must be registered. Free registration gives you access to exclusive specialist information.
Already registered? Log in here





:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ed/07/ed0756a2495cf3591db2f3c23bedd39c/0131055520v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/87/f187c86e791dfcf8ecccf615a6a3d054/0131055400v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cb/39/cb3902f5777a11e07655ba29d00a0394/0131003637v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/2e/522e60883334ab2d806e6038a11e9204/0131030069v3.jpeg)
:quality(80)/p7i.vogel.de/wcms/06/0e/060e08234d208d1faca16ad0cf582ff3/0131026470v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/6a/216a1b69d48ac4ef7fe694d4c7791531/0131025888v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4a/8d/4a8d7b8aa03d413a2bbcf34fdf463306/0131025869v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2b/5e/2b5e4798cb0d86b0f29ba4de833c1412/0131003303v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/50/5450b302b2cfe1f29c6f3d5c300123ce/0131004604v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/71/f1711b934d493949c55d6e9b9ddcffe9/0131003077v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/54/6054287023c79b93d166c8b7802f9ad4/0130972957v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f6/a8/f6a8a2ac333baa08d168cc68ac2192c0/0131001826v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/34/ff/34ff3de563d63ac821f789f8ca397070/0130971132v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e7/08/e708baaa36f8929e985dfe5baea8bc68/0130734917v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ea/5a/ea5a5ed15b2c98730c7111bb56b1a7aa/0130612537v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/50/3350efd72f2c620af7c2133ce55c901f/0131086519v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ca/fa/cafafb635ff66815b96824c2f099968a/0131058329v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/56/d7/56d73ff8d8ffc3ce50e3ff2188b7aada/0131004724v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/87/d5/87d5890059d61e8107148c2039ff9bd8/0131004713v2.jpeg)
:quality(80)/images.vogel.de/vogelonline/bdb/1710600/1710675/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1682500/1682579/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1677000/1677080/original.jpg)
:fill(fff,0)/p7i.vogel.de/companies/69/b7/69b7f63f311d9/logo-bimos-tif--2-.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/6a/216a1b69d48ac4ef7fe694d4c7791531/0131025888v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bd/aa/bdaae5b9d381e2471ab37f479ad43d79/0129143884v2.jpeg)