Ready for the Challenge? Getting accurate and reproducible trace element analysis results
Getting good and consistent results for trace element analysis can be a daily challenge. For a start, contamination lurks everywhere, ready to trip you up. Next, there is the problem of sample-to-sample washout/memory effects randomly popping up to frustrate you. Then there’s the issue of interferences, which of course are at their worst for those elements that are most important to you.
Related Vendor
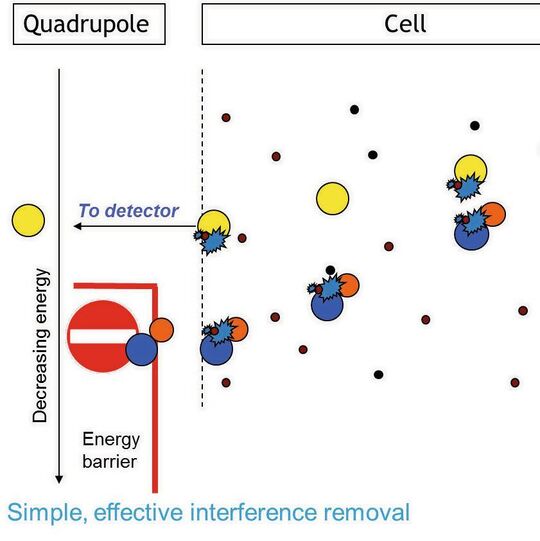
Contamination and interferences pose big challenges for getting accurate and reproducible trace element results. On top of these problems, there’s also the headache of sample and calibration solution preparation, which is not only labour intensive but also fraught with the risk of error as well as contamination. These issues are particularly prevalent for inductively coupled plasma mass spectrometry (ICP-MS) analysis, where target analytes are most often in the part per trillion to part per billion range. If you’d like to find out how to solve these problems, then read on…
Challenge 1: Contamination
Elemental contamination is a widespread issue, which encompasses contamination from sample preparation and sample-to-sample washout/memory effects (the latter appearing as cross contamination between samples). The usual suspects that cause the most trouble for trace level analysis include Al, Cr, Cu, Fe, Mn, Ni, Pb and Zn, but others, such as Hg, Mo and Sb, can cause random errors if they are at high levels in one sample, then low in the next.
During sample preparation — which includes everything from sample digestion and dilution, to blank and calibration solution preparation — the biggest sources of contamination are labware and reagents: pipette tips, volumetric flasks and their caps, sample tubes and their lids, de-ionised water supplies and reagents such as nitric acid are all potential sources of contamination.
The largest component in any liquid sample for trace element analysis using ICP-MS is most often water, so the purity of your de-ionised water supply is critical. Ideally, the water you use should have a resistivity of 18.2 MΩ cm to ensure that elemental contamination is at a minimum. The next most significant problem is surface contamination of sample bottles, volumetric flasks and sample tubes and their caps/lids. To ensure that this is eliminated, these items should be rinsed with or soaked in dilute (0.5% (v/v)) nitric acid, then washed with de-ionised water before use. Alternatively, sourcing pre-washed labware can be a good idea. Glassware generally causes more problems than plastic ware, so use plastic where possible, and recycle this as much as possible of course. Pipette tips are trickier to clean, so test tips from different suppliers to check if they are clean enough to use straight from the box. If necessary, you can also pre-clean them by pipetting dilute nitric acid and dispensing it to waste first, before using the tip for your sample preparation.
With sample-to-sample washout/memory effects, the usual troublemakers are ‘sticky’ elements such as Hg, Mo and Sb, as already mentioned. These elements take longer to washout between samples as they adhere more strongly than other elements to the surface of the sample probe and tubing along which the samples pass on their way into the ICP-MS. The simplest solution to this problem is to increase the wash time between samples, but this can lead to much longer analysis times. A mixed rinse solution containing both dilute nitric and hydrochloric acids (generally at 0.5% to 2% (v/v) concentration) will be more effective at washing out sticky elements. In the case of Hg, it is also advisable to ensure HCl is present in all blanks, calibration solutions and samples, or alternatively, to add Au at 200 ppb to all solutions to be analysed to stabilise Hg in solution and so prevent it causing washout problems.
(ID:45277001)





:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ed/07/ed0756a2495cf3591db2f3c23bedd39c/0131055520v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/87/f187c86e791dfcf8ecccf615a6a3d054/0131055400v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cb/39/cb3902f5777a11e07655ba29d00a0394/0131003637v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/2e/522e60883334ab2d806e6038a11e9204/0131030069v3.jpeg)
:quality(80)/p7i.vogel.de/wcms/06/0e/060e08234d208d1faca16ad0cf582ff3/0131026470v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/6a/216a1b69d48ac4ef7fe694d4c7791531/0131025888v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4a/8d/4a8d7b8aa03d413a2bbcf34fdf463306/0131025869v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2b/5e/2b5e4798cb0d86b0f29ba4de833c1412/0131003303v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/50/5450b302b2cfe1f29c6f3d5c300123ce/0131004604v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/71/f1711b934d493949c55d6e9b9ddcffe9/0131003077v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/54/6054287023c79b93d166c8b7802f9ad4/0130972957v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f6/a8/f6a8a2ac333baa08d168cc68ac2192c0/0131001826v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/34/ff/34ff3de563d63ac821f789f8ca397070/0130971132v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e7/08/e708baaa36f8929e985dfe5baea8bc68/0130734917v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ea/5a/ea5a5ed15b2c98730c7111bb56b1a7aa/0130612537v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/50/3350efd72f2c620af7c2133ce55c901f/0131086519v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ca/fa/cafafb635ff66815b96824c2f099968a/0131058329v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/56/d7/56d73ff8d8ffc3ce50e3ff2188b7aada/0131004724v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/87/d5/87d5890059d61e8107148c2039ff9bd8/0131004713v2.jpeg)
:quality(80)/images.vogel.de/vogelonline/bdb/1710600/1710675/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1682500/1682579/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1677000/1677080/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1434800/1434856/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1434800/1434874/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1434800/1434857/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1434800/1434858/original.jpg)
:quality(80)/p7i.vogel.de/wcms/b4/4a/b44abe3996c0f2f2c2e40a23f6a67e1f/0125818584v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8f/66/8f66b8acbb3037ca8e6b320bcad1c817/0129118043v2.jpeg)