Switzerland: Diabetes Research Can Cells Change Their Identity?
By observing how cells manage to modify their function by partially changing their identity, Swiss researchers discovered a phenomenon of cell plasticity unknown until now. Although they only examined cells in the pancreas, such processes might characterize many other cell types in the body.
Related Vendors
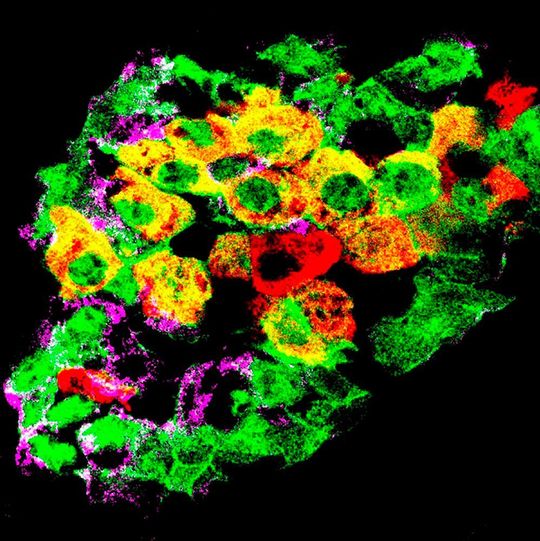
Geneva/Switzerland — Diabetes is characterized by persistent high blood sugar levels that occur when certain cells in the pancreas — the insulin-producing β cells — are destroyed or are no longer able to secrete insulin. Researchers at the University of Geneva (Unige) have succeeded in showing how part of the pancreatic α and δ cells, which usually produce other hormones, can take over from the damaged β cells by starting to produce insulin. These results, to be read in Nature Cell Biology, lead to envision entirely new therapeutic strategies that could harness the body’s own regenerative capacities.
The pancreas contains several types of cells that produce different hormones responsible for regulating blood sugar levels. Among these are the α cells, which produce glucagon, the β cells, which produce insulin, and the δ cells, which produce somatostatin, a hormone that acts as a local regulator to control the activity of the δ and β cells. Together, they form small clusters known as pancreatic islets. Glucagon increases blood sugar levels, while insulin has the opposite effect. In patients with diabetes, in the absence of functional β cells, blood sugar levels are persistently high.
At the Unige Faculty of Medicine, Professor Pedro Herrera and his team had, a few years ago, demonstrated the existence of a natural capacity to regenerate insulin-producing cells: in mice without β cells, new insulin-producing cells appear spontaneously. Some pancreatic cells are thus able to reprogram themselves to learn how to secrete insulin. “However, this phenomenon only concerns 1 to 2 % of α and δ cells. Why do some cells do this conversion and others not? And above all, would it be possible to encourage it? These are the questions that are at the heart of our work,” explains Professor Herrera.
A Modification of Local Origin
Scientists first conducted gene expression analyses before and after the disappearance of β cells. They observed, in α cells, modifications that appear to go in opposite directions: the overexpression of certain genes typical of insulin-producing β cells, but also, and at the same time, that of certain genes typical of glucagon-producing α cells. The normal functioning of α cells is linked to the action of insulin, as shown by the presence of insulin receptors on their surface. Hence, α cell activity is disrupted when β cells are destroyed.
:quality(80)/images.vogel.de/vogelonline/bdb/1460300/1460362/original.jpg)
Japan: New Cell Culture Platform
Natural Behavior of Cancer Cells Revealed
But what signal induces cell conversion? To explore different possibilities, the researchers transplanted pancreatic islets into normal mice. First hypothesis: hyperglycaemia. In transplanted but non-diabetic mice, whose β cells remain perfectly functional and do not have hyperglycaemia, some α cells of the graft began to produce insulin when the β cells died in the transplanted islets. Consequently, hyperglycaemia does not cause α cell reallocation. The cellular pancreatic environment surrounding the islets is not involved either, since α cell conversion in grafts occurred in the kidney capsule, i.e. outside of the pancreas. The only explanation is that the reprogramming capacity is intrinsic to the very pancreatic islet where these cells are located. “Thus, in the same graft, only islets without β cells displayed reprogramming. No cell conversion occurred in neighbouring islets containing all their β cells,” says Pedro Herrera.
Disturbed Signals
Continuing their study, the researchers blocked, in non-diabetic mice, the receptors located on α cells surface to detect the presence of insulin. Some of these α cells then began to produce insulin, indicating that insulin itself would act as a brake, preventing the reprogramming of α cells. “By administering an insulin antagonist drug, we were able to increase the number of α cells that started producing insulin by 1 to 5 %. In doing so, these cells became hybrids: they partially, but not fully changed their identity, and the phenomenon was reversible depending on the circumstances influencing the cells. Now that we are beginning to understand the mechanisms of this cellular plasticity, we believe that these adaptive cell identity changes could be exploited in future new treatments.”
:quality(80)/images.vogel.de/vogelonline/bdb/1441400/1441486/original.jpg)
Germany: Cell Biology
3D Analysis of Cells During Organ Growth
Other Organs Should Have the Same Plasticity Potential
If Professor Herrera’s work focuses on pancreatic cells, the same processes could apply to many other differentiated cells in the body. Thus, the idea that differentiated functional mature cells remain stable forever is now being challenged. “What we are showing here is that the state of differentiation of a given cell is not carved in stone. Cell identity, at all stages of life, is modulated by the immediate cellular environment, particularly by inhibitory signals. Cell identity maintenance is therefore an active process of inhibition throughout the life of the cell, and not an intrinsic or passive state of differentiation. This ability of specialized cells to change their function could prove crucial for treating other pathologies that are due to massive or inappropriate cell death, such as Alzheimer’s disease or myocardial infarction,” concludes Professor Herrera.
(ID:45565567)





:quality(80)/p7i.vogel.de/wcms/d6/25/d625408f8628b90bf08a73d0c6d57fe7/0131163821v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/0e/72/0e7296d4ca7d79eb7b3e4f668641af8f/0131148508v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ed/07/ed0756a2495cf3591db2f3c23bedd39c/0131055520v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/2e/522e60883334ab2d806e6038a11e9204/0131030069v3.jpeg)
:quality(80)/p7i.vogel.de/wcms/06/0e/060e08234d208d1faca16ad0cf582ff3/0131026470v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/6a/216a1b69d48ac4ef7fe694d4c7791531/0131025888v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4a/8d/4a8d7b8aa03d413a2bbcf34fdf463306/0131025869v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/23/e2/23e2e753cc718480ede0fadd24e0ee02/0131121023v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b5/63/b5634bbb6ac68f7f55fb261344d6fa1f/0131163645v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2b/5e/2b5e4798cb0d86b0f29ba4de833c1412/0131003303v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/50/5450b302b2cfe1f29c6f3d5c300123ce/0131004604v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/48/97/4897129d96e4ab9287933406b153ab51/0131163956v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f6/a8/f6a8a2ac333baa08d168cc68ac2192c0/0131001826v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/34/ff/34ff3de563d63ac821f789f8ca397070/0130971132v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e7/08/e708baaa36f8929e985dfe5baea8bc68/0130734917v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/25/d8/25d8dd47da6179f23914adb6bb448757/0131194053v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/df/db/dfdb82e64a787f853ed7159809c80f3c/0131142428v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/50/3350efd72f2c620af7c2133ce55c901f/0131086519v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ca/fa/cafafb635ff66815b96824c2f099968a/0131058329v2.jpeg)
:quality(80)/images.vogel.de/vogelonline/bdb/1710600/1710675/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1682500/1682579/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1677000/1677080/original.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/131900/131910/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/69/b7/69b7f63f311d9/logo-bimos-tif--2-.jpeg)
:quality(80)/p7i.vogel.de/wcms/7f/75/7f7588857d84065177747494c48b89c6/0128725945v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/ae/75/ae754d5c886d219a0746f2855f433746/0128230215v2.jpeg)