China: Rheumatism Drug 3D-Printed Tablet Releases Active Substance in a Controlled Way
Chinese pharmaceutical company Triastek specializes in 3D printed drugs. Now, its Investigational New Drug (IND) 505(b)(2) application for its first 3D-printed drug, T19, has been granted for use by the US Food and Drug Administration.
Related Vendors

Nanjing/China — T19 was developed for the treatment of rheumatoid arthritis (RA). The 3D-printed design enables the tablet to function like a chronotherapeutic drug delivery system, acting on the circadian rhythm of RA symptoms. Patients take the drug before bedtime, and the active ingredient in the tablet is released in a delayed manner, so blood concentrations are highest in the early morning hours, when symptoms such as pain, joint stiffness and dysfunction are most acute.
The Melt Extrusion Deposition(MED) Technology
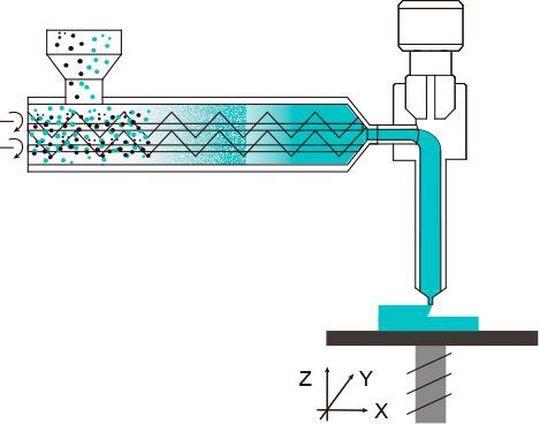
Using Melt Extrusion Deposition (MED) 3D printing technology, the T19 tablet structures have a three-dimensional shape that allows precise control of drug release. In the MED process, powdered raw materials and additives are first mixed, then melted, and then continuously extruded layer by layer. Complex structures and the combination of active compounds can be achieved by increasing the number of nozzles.
Intelligent Pharmaceutical Manufacturing
The MED 3D printing technology platform includes digital design of the pharmaceutical dosage form, an efficient product development approach and automated smart manufacturing. According to Triastek, this allows tablets to be designed with more sophisticated shapes and internal geometric structures that can be used to modulate the onset of action, kinetics, duration and mode of drug release with great predictability and reproducibility. These customized release kinetics can contribute to enhancement of therapeutic effect, reduction of side effects, improvement of compliance, and efficient development of pharmaceutical products at various stages.
Digital Formulation Development
Furthermore, Triastek has developed a novel pharmaceutical product development methodology, 3D Printing Formulation by Design (3DFbD), to bypass the trial and error or experience-based conventional formulation development process. This methodology significantly improves the efficiency and success rate of drug product development, thereby reducing development time and costs. In addition, the integration of real-time process analytical technology (PAT) into the MED continuous 3D printing system enables consistent monitoring of the manufacturing process to ensure production quality, reduce manufacturing costs, and facilitate regulatory oversight.
IND Approval of T19 Is Major Milestone
In April 2020, Triastek's 3D printing via MED was accepted into the FDA's Emerging Technology Program (ETP). The FDA accepted the MED 3D printing process based on the following features:
- provided use of MED-based 3D technology to produce a modified release solid oral dosage form
- fully automated process with PAT and feedback controls.
- Triastek plans to file for IND approval in China later this year. Additional applications in Japan and Europe are expected to follow. The New Drug Application (NDA) for T19 is expected to be submitted to the U.S. FDA in 2023.
This article was first published by Mission Additive
(ID:47150869)





:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ed/07/ed0756a2495cf3591db2f3c23bedd39c/0131055520v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/87/f187c86e791dfcf8ecccf615a6a3d054/0131055400v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cb/39/cb3902f5777a11e07655ba29d00a0394/0131003637v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/2e/522e60883334ab2d806e6038a11e9204/0131030069v3.jpeg)
:quality(80)/p7i.vogel.de/wcms/06/0e/060e08234d208d1faca16ad0cf582ff3/0131026470v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/6a/216a1b69d48ac4ef7fe694d4c7791531/0131025888v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4a/8d/4a8d7b8aa03d413a2bbcf34fdf463306/0131025869v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2b/5e/2b5e4798cb0d86b0f29ba4de833c1412/0131003303v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/50/5450b302b2cfe1f29c6f3d5c300123ce/0131004604v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/71/f1711b934d493949c55d6e9b9ddcffe9/0131003077v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/54/6054287023c79b93d166c8b7802f9ad4/0130972957v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f6/a8/f6a8a2ac333baa08d168cc68ac2192c0/0131001826v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/34/ff/34ff3de563d63ac821f789f8ca397070/0130971132v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e7/08/e708baaa36f8929e985dfe5baea8bc68/0130734917v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ea/5a/ea5a5ed15b2c98730c7111bb56b1a7aa/0130612537v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/50/3350efd72f2c620af7c2133ce55c901f/0131086519v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ca/fa/cafafb635ff66815b96824c2f099968a/0131058329v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/56/d7/56d73ff8d8ffc3ce50e3ff2188b7aada/0131004724v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/87/d5/87d5890059d61e8107148c2039ff9bd8/0131004713v2.jpeg)
:quality(80)/images.vogel.de/vogelonline/bdb/1710600/1710675/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1682500/1682579/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1677000/1677080/original.jpg)
:fill(fff,0)/p7i.vogel.de/companies/5f/3f/5f3f63ceedc78/knf-logo.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/131900/131910/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/69/b7/69b7f63f311d9/logo-bimos-tif--2-.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/eb/1deb14647220ec214951d03f426ad28d/0130642607v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8f/b9/8fb98a958e4afe679865fbe52a473680/0129940982v2.jpeg)